For US Health Care
Professionals Only

Patients who undergo an alloHCT can develop complications, such as debilitating and life-threatening cGVHD.1-3 In fact, cGVHD is the leading cause of nonrelapse mortality in patients surviving >2 years post alloHCT.3
Risk factors for identifying patients who are more likely to develop cGVHD4-7
Of patients who received ≥3 lines of systemic therapy, 42% had involvement of ≥4 organs at the time of diagnosis.8
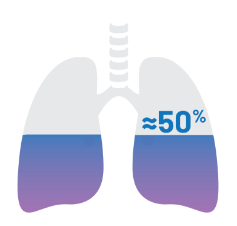
Treatment of fibrosis in the lungs is a critical unmet need.10-13
Find out the difference REZUROCK can make.
Treatment of fibrosis in the lungs is a critical unmet need.10-13
The relationship between inflammation and fibrosis in cGVHD is complex and not fully understood.14,15 However, there is an association between chronic inflammatory changes in cGVHD and collagen-producing fibroblasts.16,17 This can lead to the development of fibrotic lesions across multiple organs.16,18
REZUROCK is an
designed for the treatment of cGVHD.27-29
Explore the rockstar studyaGVHD, acute graft-versus-host disease; alloHCT, allogeneic hematopoietic cell transplant; ATG, antithymocyte globulin; cGVHD, chronic graft-versus-host disease; CMV, cytomegalovirus; GI, gastrointestinal; HLA, human leukocyte antigen; MOA, mechanism of action; RIC, reduced-intensity conditioning.
References: 1. Arora M, Pidala J, Cutler CS, et al. Impact of prior acute GVHD on chronic GVHD outcomes: a Chronic Graft versus Host Disease Consortium study. Leukemia. 2013;27(5):1196-1201. doi:10.1038/leu.2012.292 2. Wingard JR, Majhail NS, Brazauskas R, et al. Long-term survival and late deaths after allogeneic hematopoietic cell transplantation. J Clin Oncol. 2011;29(16):2230-2239. doi:10.1200/JCO.2010.33.7212 3. Arai S, Arora M, Wang T, et al; for the Graft-vs-Host Disease Working Committee of the CIBMTR. Increasing incidence of chronic graft-versus-host disease in allogeneic transplantation: a report from the Center for International Blood and Marrow Transplant Research. Biol Blood Marrow Transplant. 2015;21(2):266-274. doi:10.1016/j.bbmt.2014.10.021 4. Afram G, Pérez Simón JA, Remberger M, et al. Reduced intensity conditioning increases risk of severe cGVHD: identification of risk factors for cGVHD in a multicenter setting. Med Oncol. 2018;35(6):79. doi:10.1007/s12032-018-1127-2 5. Chen Y-B, Wang T, Hemmer MT, et al. GvHD after umbilical cord blood transplantation for acute leukemia: an analysis of risk factors and effect on outcomes. Bone Marrow Transplant. 2017;52(3):400-408. doi:10.1038/bmt.2016.265 6. Lazaryan A, Weisdorf DJ, DeFor T, et al. Risk factors for acute and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation with umbilical cord blood and matched sibling donors. Biol Blood Marrow Transplant. 2016;22(1):134-140. doi:10.1016/j.bbmt.2015.09.008 7. Kok LMC, Bungener L, de Bock GH, et al. Risk factors associated with the development of moderate to severe chronic graft-versus-host disease after non-myeloablative conditioning allogeneic stem cell transplantation in patients with AML or MDS. Hum Cell. 2020;33(1):243-251. doi:10.1007/s13577-019-00297-7 8. Data on file. Kadmon Pharmaceuticals, LLC; 2018. 9. Jacobsohn DA, Kurland BF, Pidala J, et al. Correlation between NIH composite skin score, patient-reported skin score, and outcome: results from the Chronic GVHD Consortium. Blood. 2012;120(13):2545-2552. doi:10.1182/blood-2012-04-424135 10. Arora M, Cutler CS, Jagasia MH, et al. Late acute and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2016;22(3):449-455. doi:10.1016/j.bbmt.2015.10.018 11. Inagaki J, Moritake H, Nishikawa T, et al. Long-term morbidity and mortality in children with chronic graft-versus-host disease classified by National Institutes of Health consensus criteria after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2015;21(11):1973-1980. doi:10.1016/j.bbmt.2015.07.025 12. Inamoto Y, Martin PJ, Chai X, et al; on behalf of the Chronic GVHD Consortium. Clinical benefit of response in chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2012;18(10):1517-1524. doi:10.1016/j.bbmt.2012.05.016 13. Gazourian L, Spring L, Meserve E, et al. Pulmonary clinicopathological correlation after allogeneic hematopoietic stem cell transplantation: an autopsy series. Biol Blood Marrow Transplant. 2017;23(10):1767-1772. doi:10.1016/j.bbmt.2017.06.009 14. Kitko CL, White ES, Baird K. Fibrotic and sclerotic manifestations of chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2012;18(1 suppl):S46-S52. doi:10.1016/j.bbmt.2011.10.021 15. Cooke KR, Luznik L, Sarantopoulos S, et al. The biology of chronic graft-versus-host disease: a task force report from the National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease. Biol Blood Marrow Transplant. 2017;23(2):211-234. doi:10.1016/j.bbmt.2016.09.023 16. MacDonald KPA, Hill GR, Blazar BR. Chronic graft-versus-host disease: biological insights from preclinical and clinical studies. Blood. 2017;129(1)13-21. doi:10.1182/blood-2016-06-686618 17. Zeiser R, Blazar BR. Pathophysiology of chronic graft-versus-host disease and therapeutic targets. N Engl J Med. 2017;377(26):2565-2579. doi:10.1056/NEJMra1703472 18. Fiuza-Luces C, Simpson RJ, Ramírez M, Lucia A, Berger NA. Physical function and quality of life in patients with chronic GvHD: a summary of preclinical and clinical studies and a call for exercise intervention trials in patients. Bone Marrow Transplant. 2016;51(1):13-26. doi:10.1038/bmt.2015.195 19. Henden AS, Hill GR. Cytokines in graft-versus-host disease. J Immunol. 2015;194(10):4604-4612. doi:10.4049/jimmunol.1500117 20. Chronic graft-vs-host disease of skin and connective tissues. BMT InfoNet. Accessed April 19, 2022. Bmtinfonet.org/video/chronic-graft-vs-host-disease-skin-and-connective-tissues 21. Cutler CS, Koreth J, Ritz J. Mechanistic approaches for the prevention and treatment of chronic GVHD. Blood. 2017;129(1):22-29. doi:10.1182/blood-2016-08-686659 22. Jakafi. Package insert. Incyte Corporation; September 2021. 23. Modi B, Hernandez-Henderson M, Yang D, et al. Ruxolitinib as salvage therapy for chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2019;25(2):265-269. doi:10.1016/j.bbmt.2018.09.003 24. Imbruvica. Package insert. Pharmacyclics LLC; 2020. 25. Hill L, Alousi A, Kebriaei P, Mehta R, Rezvani K, Shpall E. New and emerging therapies for acute and chronic graft versus host disease. Ther Adv Hematol. 2018;9(1):21-46. doi:10.1177/2040620717741860 26. Koreth J, Kim HT, Jones KT, et al. Efficacy, durability, and response predictors of low-dose interleukin-2 therapy for chronic graft-versus-host disease. Blood. 2016;128(1):130-137. doi:10.1182/blood-2016-02-702852 27. REZUROCK. Package insert. Kadmon Pharmaceuticals, LLC; 2023. 28. Zanin-Zhorov A, Weiss JM, Nyuydzefe MS, et al. Selective oral ROCK2 inhibitor down-regulates IL-21 and IL-17 secretion in human T cells via STAT3-dependent mechanism. Proc Natl Acad Sci USA. 2014;111(47):16814-16819. doi:10.1073/pnas.1414189111 29. Flynn R, Paz K, Du J, et al. Targeted Rho-associated kinase 2 inhibition suppresses murine and human chronic GVHD through a Stat3-dependent mechanism. Blood. 2016;127(17):2144-2154. doi:10.1182/blood-2015-10-678706