For US Health Care
Professionals Only


These patients had
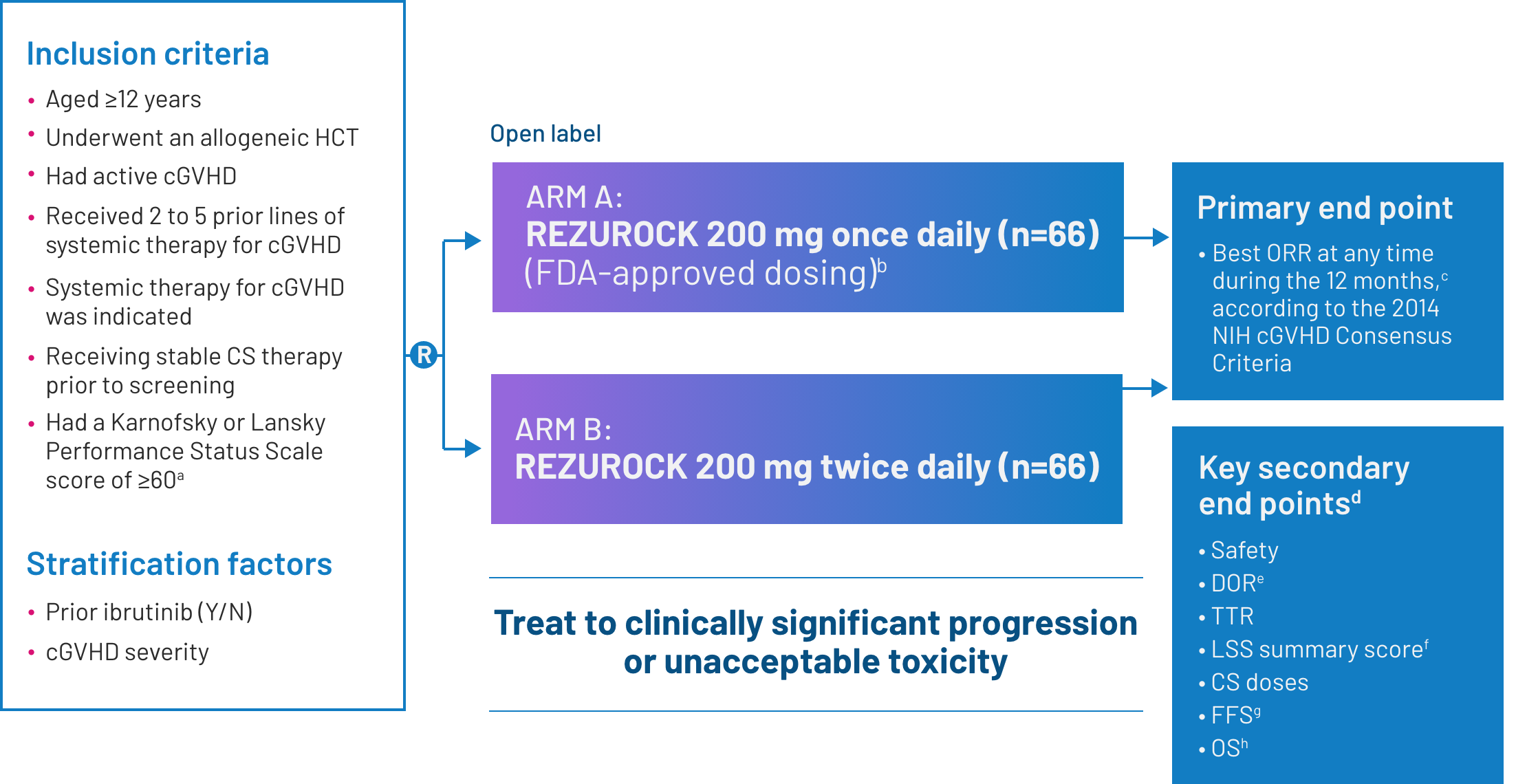
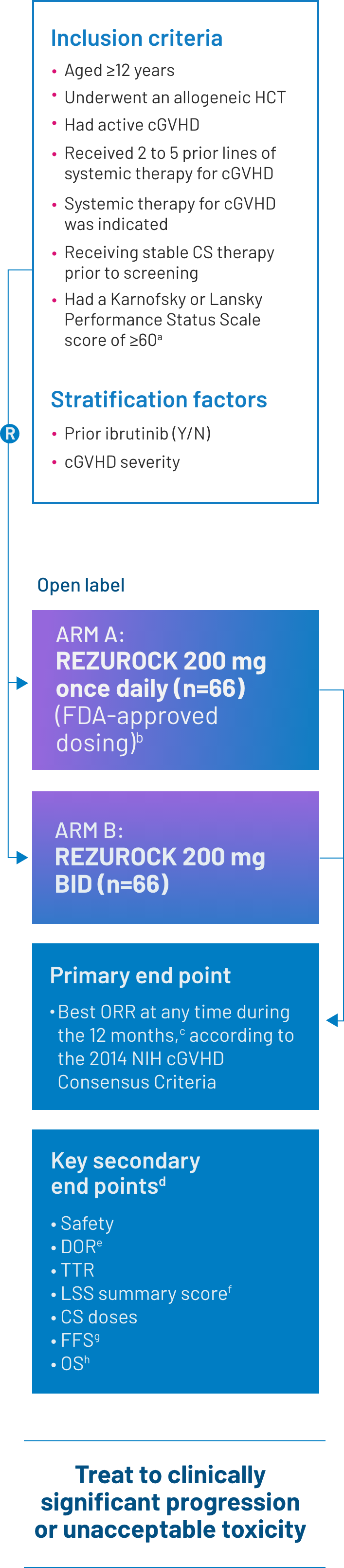
ROCKstar was a pivotal phase 2, open-label, randomized, multicenter study that evaluated the efficacy and safety of REZUROCK in patients with cGVHD after receiving 2 to 5 prior lines of systemic therapy.2
Exclusion criteria: Patients were excluded from the study if they had a relapse of their underlying malignancy, had developed posttransplant lymphoproliferative disease, were currently receiving ibrutinib2 or had any of the following laboratory results: platelets were < 50 × 109/L; absolute neutrophil count < 1.5 × 109/L; AST or ALT > 3 × ULN; total bilirubin > 1.5 × ULN; QTc(F) >480 ms; eGFR <30 mL/min/1.73 m2; or FEV1 ≤39%.1
Screening for eligibility was conducted within 14 days of C1D1.2
Certain concurrent immunosuppressive medications were allowed, as drug-drug interactions were not anticipated.2
aThe Karnofsky/Lansky Performance Status Scale is used in outcome-based analyses to determine the functional status of a patient. The Karnofsky Scale is designed for patients aged ≥16 years, whereas the Lansky Scale is designed for patients aged ≤16 years. The scale range for both is 10 to 100.3
bThe final FDA interpretation of the ROCKstar study omitted 1 patient from the REZUROCK 200-mg once-daily arm. As a result, there are minor differences between the ROCKstar publication, where n=66, and the Prescribing Information, where n=65.
cORR was defined as the proportion of patients who achieved CR or PR. All responses were assessed by the study site investigators.1
dPrespecified secondary end points; not powered to show statistical significance.
eDOR was measured from the time of initial PR or CR until documented progression from best response of cGVHD, time from initial response to start of additional systemic cGVHD therapy or death.2
fThe 7-day LSS summary score was calculated based on the developer recommendations and was compared with the score from baseline in an exploratory analysis. An improvement of ≥7 points was considered clinically meaningful.2
gFFS was defined as the absence of relapse, nonrelapse mortality or a need for additional systemic therapy.2
hOS was defined as the time from the first dose of REZUROCK to the date of death due to any cause.4
The baseline demographics demonstrate the diversity of the patient population in the ROCKstar study. Many patients presented with challenging characteristics, including advanced or complex disease and having received multiple prior lines of systemic therapy.
| Characteristics | REZUROCK 200 mg once daily (n=66)b |
| Median age, y (range) | 53 (21-77) |
| Male, n (%) | 42 (64) |
| Median prior lines of systemic therapy, n | 3 |
| Median time from cGVHD diagnosis to enrollment, mo (range) | 25 (2-162) |
| Median prednisone-equivalent dose at enrollment, mg/kg/d (range) | 0.20 (0.03-0.95) |
| Concomitant PPI use, n (%) | 33 (50) |
| ≥4 organs involved, n (%) | 33 (50) |
| Previous aGVHD, n (%) | 42 (64) |
| Refractory to prior line of systemic therapy, n (%i) | 44 (79) |
| NIH-defined cGVHD severity, n (%) | |
| Severe | 46 (70) |
| Moderate | 18 (27) |
| Mild | 2 (3) |
| Prior systemic cGVHD therapy type, n (%) | |
| CS (prednisone) | 65 (99) |
| Tacrolimus | 40 (61) |
| ECP | 31 (47) |
| Sirolimus | 29 (44) |
| Ibrutinib | 22 (33) |
| Ruxolitinib | 20 (30) |
| Cyclosporine | 4 (6) |
| Imatinib | 3 (5) |
bThe final FDA interpretation of the ROCKstar study omitted 1 patient from the REZUROCK 200-mg once-daily arm. As a result, there are minor differences between the ROCKstar publication, where n=66, and the Prescribing Information, where n=65.
iDenominator excludes patients with unknown status.1
bThe final FDA interpretation of the ROCKstar study omitted 1 patient from the REZUROCK 200-mg once-daily arm. As a result, there are minor differences between the ROCKstar publication, where n=66, and the Prescribing Information, where n=65.
| Organ involvement | Patients (n=66),b n (%) |
| Eyes | 48 (73) |
| Skin | 55 (83) |
| Mouth | 30 (46) |
| Joints/Fascia | 51 (77) |
| Lungs | 24 (36) |
| Upper GI | 13 (20) |
| Esophagus | 19 (29) |
| Lower GI | 6 (9) |
| Liver | 9 (14) |
bThe final FDA interpretation of the ROCKstar study omitted 1 patient from the REZUROCK 200-mg once-daily arm. As a result, there are minor differences between the ROCKstar publication, where n=66, and the Prescribing Information, where n=65.
aGVHD, acute graft-versus-host disease; ALT, alanine aminotransferase; AST, aspartate aminotransferase; cGVHD, chronic graft-versus-host disease; C1D1, cycle 1 day 1; CNI, calcineurin inhibitor; CR, complete response; CS, corticosteroid(s); DOR, duration of response; ECP, extracorporeal photopheresis; eGFR, estimated glomerular filtration rate; FDA, US Food and Drug Administration; FEV1, forced expiratory volume in 1 second; FFS, failure-free survival; GI, gastrointestinal; HCT, hematopoietic cell transplant; LFT, liver function test; LSS, Lee Symptom Scale; mITT, modified intent-to-treat; MOA, mechanism of action; NIH, National Institutes of Health; NRM, nonrelapse mortality; ORR, overall response rate; OS, overall survival; PPI, proton pump inhibitor; PR, partial response; QOL, quality of life; TTR, time to response; ULN, upper limit of normal.
References: 1. REZUROCK. Package insert. Kadmon Pharmaceuticals, LLC; 2023. 2. Cutler C, Lee SJ, Arai S, et al; on behalf of the ROCKstar Study Investigators. Belumosudil for chronic graft-versus-host disease after 2 or more prior lines of therapy: the ROCKstar Study. Blood. 2021;138(22):2278-2289. doi:10.1182/blood.2021012021 3. Center for International Blood and Marrow Transplant Research. Karnofsky/Lansky performance status. Accessed April 20, 2022. https://www.cibmtr.org/DataManagement/TrainingReference/Manuals/DataManagement/Documents/appendix-l.pdf 4. Data on file. Kadmon Pharmaceuticals, LLC; 2021.