For US Health Care
Professionals Only



Clinically meaningful and statistically significant ORRa in a real-world demographic of patients1

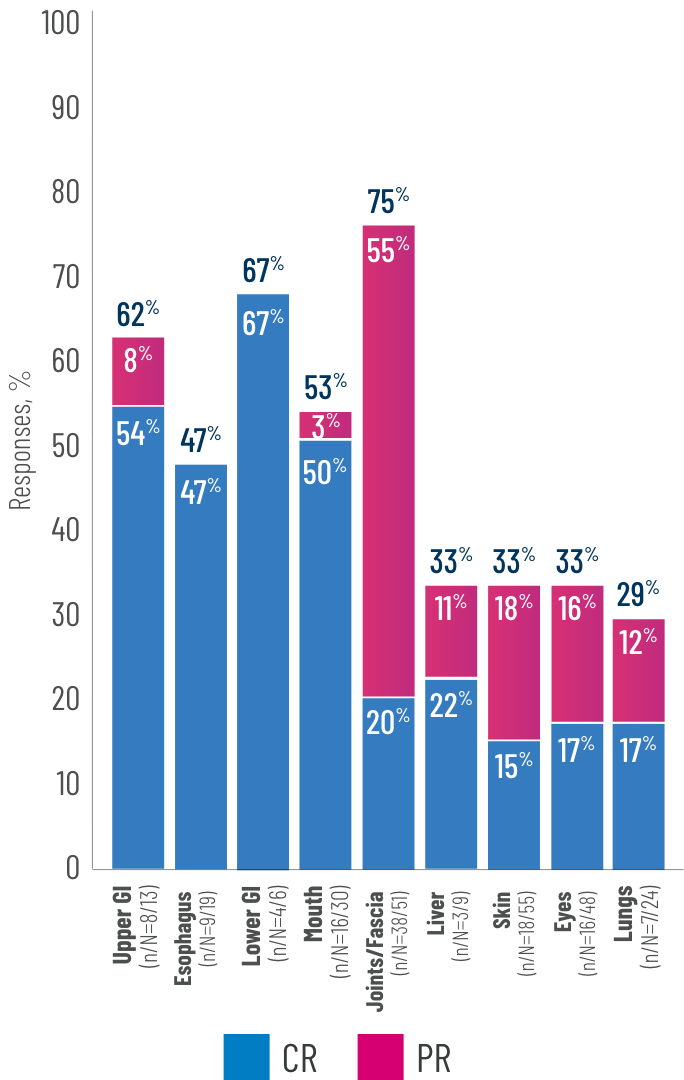
Clinically meaningful ORRs observed across key subgroups2

Responses across all evaluated organs, including those with fibrotic manifestations2

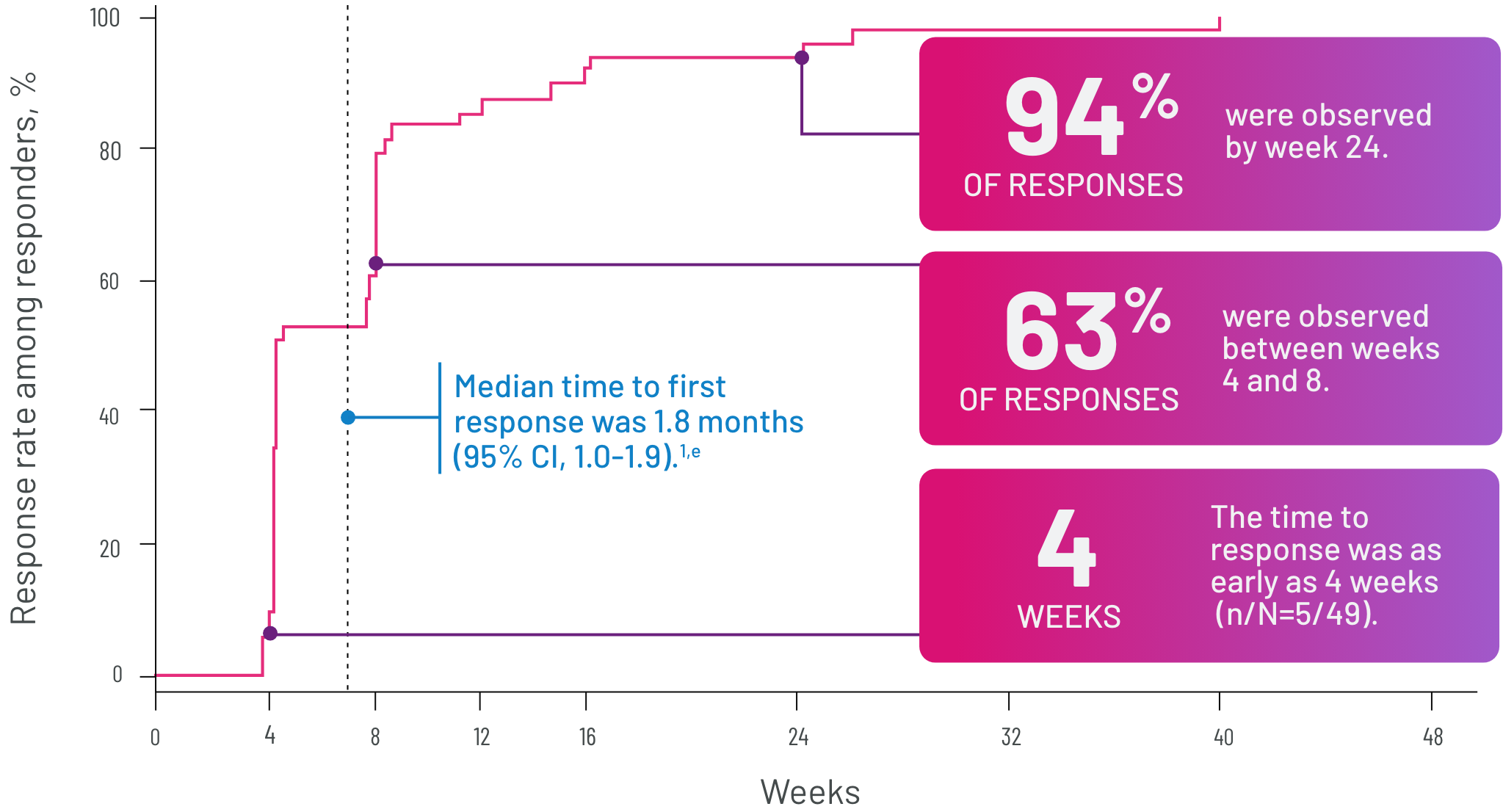
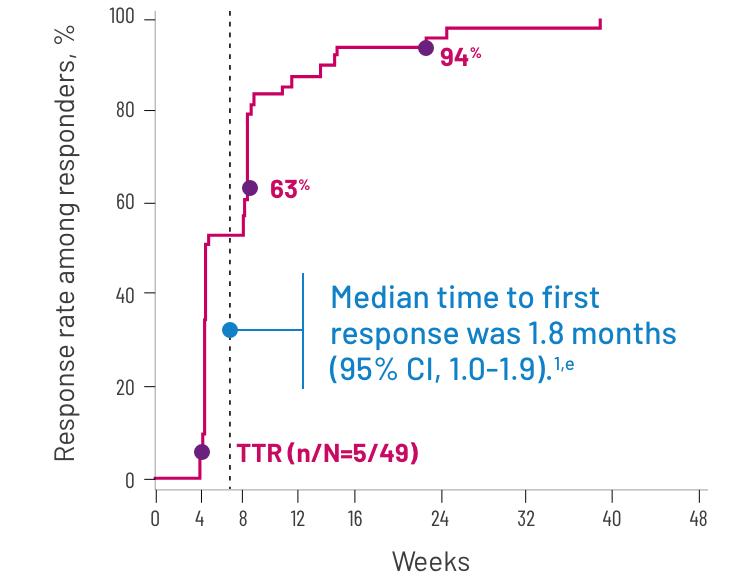
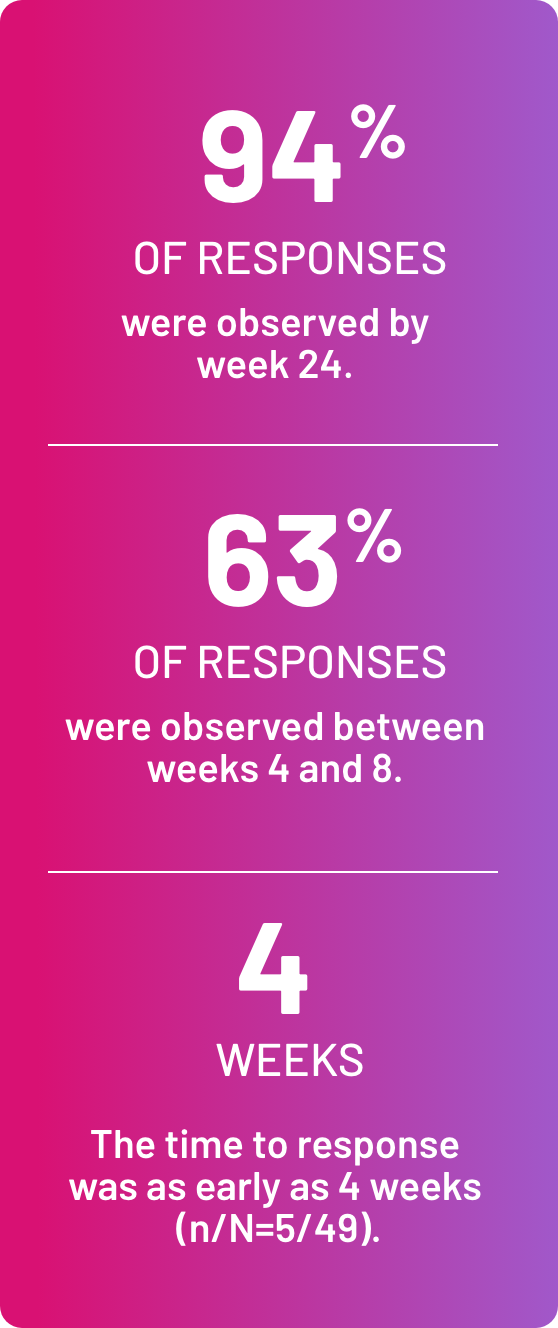
Most responses were seen between 4 and 8 weeks3

FFSb rate at 6 and 12 months and OSc rate at 24 months2,3

Exploratory analyses of LSSd summary score showed clinically meaningful improvements (≥7-point reduction) in patient-reported QOL1

Reduction in use of CS and CNI therapies2,3
aProportion of patients who achieved CR or PR according to the 2014 NIH cGVHD Consensus Criteria.1
bFFS was defined as the absence of relapse, nonrelapse mortality or a need for additional systemic therapy.2
cOS was defined as the time from the first dose of REZUROCK to the date of death due to any cause.3
dThe LSS is a 30-item, 7-subscale symptom scale and QOL measurement tool that evaluates the AEs of cGVHD in the categories of skin, vitality, lung, nutritional status, psychological functioning, eye and mouth.4

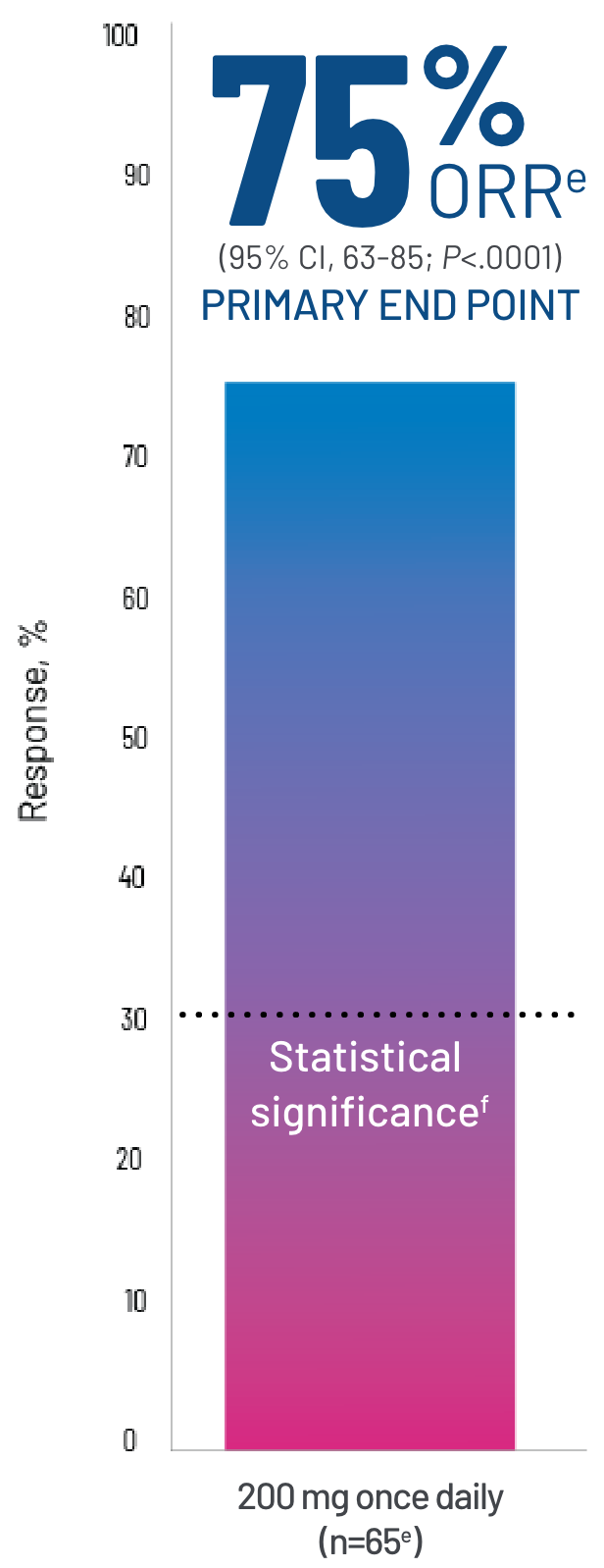
CR, n=4 (6%). PR, n=45 (69%).1
aProportion of patients who achieved CR or PR according to the 2014 NIH cGVHD Consensus Criteria.1
eBased on a final analysis by the FDA (n=65).
fStatistical significance was achieved if the lower bound of the 95% CI of ORR exceeded 30%.5
Defined as <28 months from initial diagnosis.
(n/N=32/36)
(n/N=35/46)
(n/N=24/33)
(n/N=21/30)
(n/N=9/12)
(n/N=16/22)
(n/N=13/20)
did not experience death or new systemic therapy initation (95% CI, 46-74) for ≥12 months after response.1
eBased on a final analysis by the FDA (n=65).
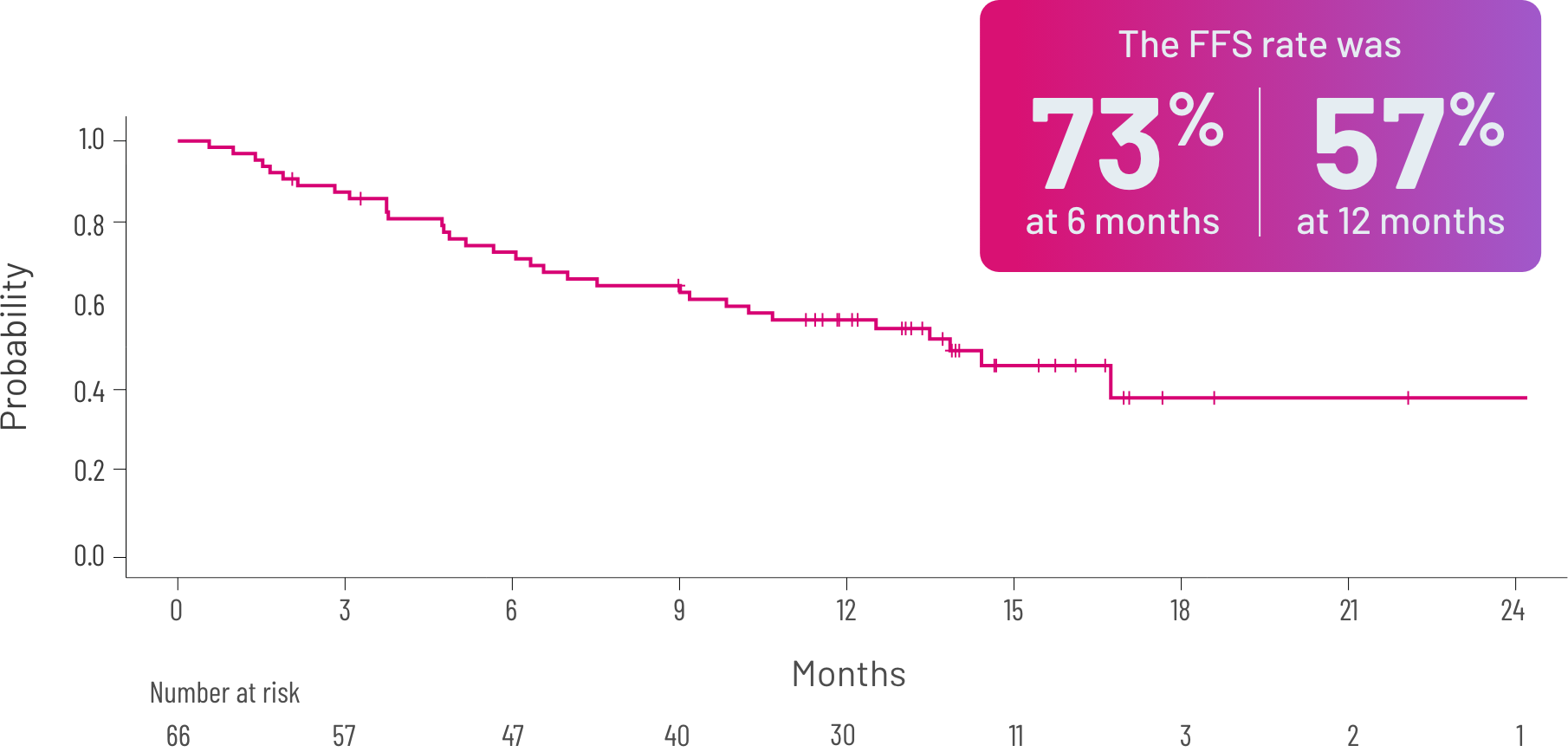
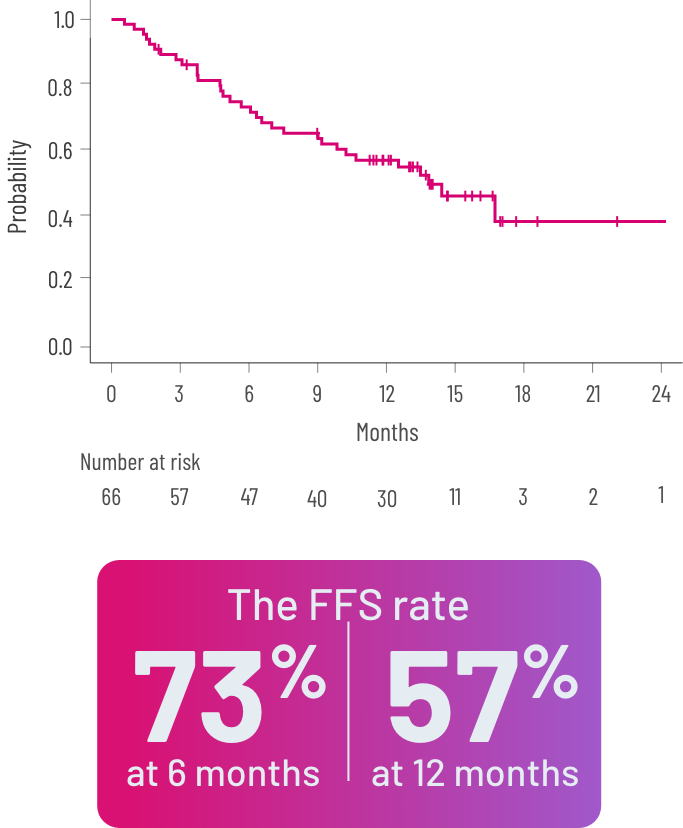
Kaplan-Meier curve of estimated FFS.
bFFS was defined as the absence of relapse, nonrelapse mortality or a need for additional systemic therapy.2
gPrespecified secondary end point; not powered to show statistical significance.
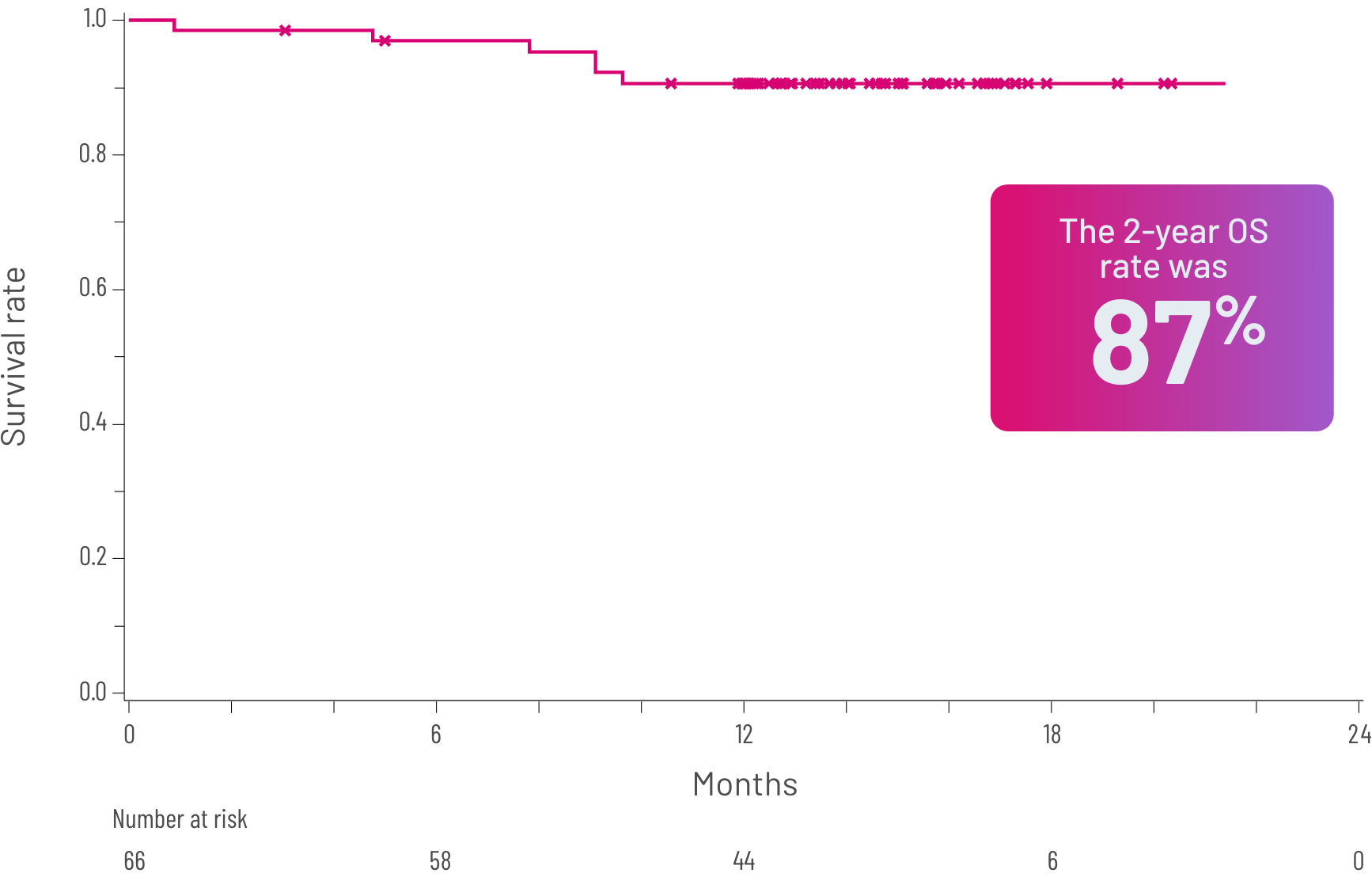
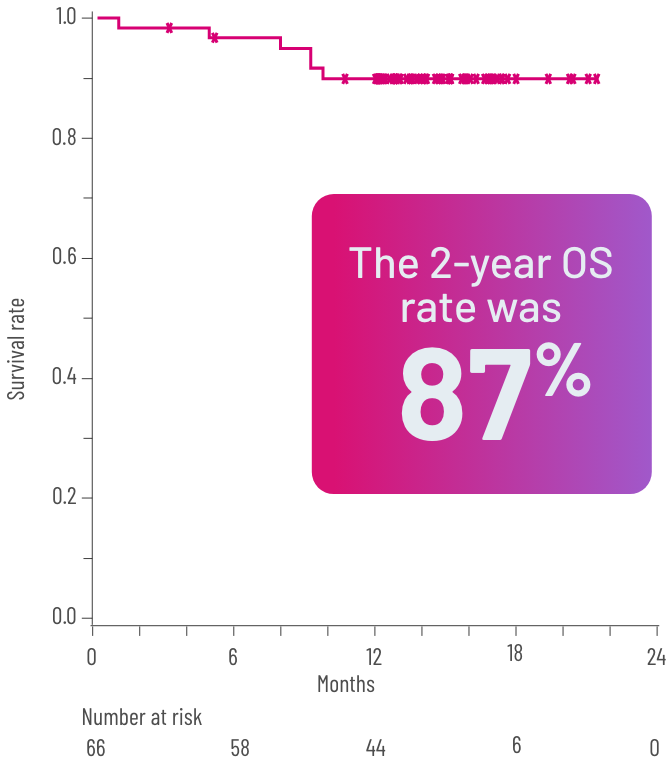
Kaplan-Meier curve of estimated OS.
cOS was defined as the time from the first dose of REZUROCK to the date of death due to any cause.3
gPrespecified secondary end point; not powered to show statistical significance.
(≥7-point reduction in LSSd summary score) with REZUROCK 200 mg once daily in the mITT population in an exploratory analysis
(≥7-point reduction in LSSd summary score) with REZUROCK 200 mg once daily in the mITT population in an exploratory analysis
Both responders (61%) and nonresponders (25%) had improved QOL scores.7
dThe LSS is a 30-item, 7-subscale symptom scale and QOL measurement tool that evaluates the AEs of cGVHD in the categories of skin, vitality, lung, nutritional status, psychological functioning, eye and mouth.4
eBased on a final analysis by the FDA (n=65).
hThrough cycle 7 day 1.1
52%OF PATIENTSe REPORTED IMPROVEMENTS IN QOL(95% CI, 40-65) |
CLINICALLY MEANINGFUL IMPROVEMENTS IN PATIENT-REPORTED QOL1,h(≥7-point reduction in LSSd summary score) with REZUROCK 200 mg once daily in the mITT population in an exploratory analysis
dThe LSS is a 30-item, 7-subscale symptom scale and QOL measurement tool that evaluates the AEs of cGVHD in the categories of skin, vitality, lung, nutritional status, psychological functioning, eye and mouth.4 eBased on a final analysis by the FDA (n=65). hThrough cycle 7 day 1.1 |
of patients (n=42).
in patients.
of patients (n=13).
of patients (n=10).
of patients (n=4).
eBased on a final analysis by the FDA (n=65).
gPrespecified secondary end point; not powered to show statistical significance.
iNonresponders were defined as patients with CR or PR in ≥1 organ, accompanied by progression in another organ (considered progression); outcomes that did not meet the criteria for CR, PR, progression or mixed response; or progression in ≥1 organ or site without a response in any other organ or site.5
AE, adverse event; cGVHD, chronic graft-versus-host disease; CNI, calcineurin inhibitor; CR, complete response; CS, corticosteroid; DOR, duration of response; FDA, US Food and Drug Administration; FFS, failure-free survival; GI, gastrointestinal; LSS, Lee Symptom Scale; mITT, modified intent-to-treat; MOA, mechanism of action; ORR, overall response rate; OS, overall survival; PR, partial response; QOL, quality of life; TTR, time to response.
References: 1. REZUROCK. Package insert. Kadmon Pharmaceuticals, LLC; 2023. 2. Cutler C, Lee SJ, Arai S, et al; on behalf of the ROCKstar Study Investigators. Belumosudil for chronic graft-versus-host disease after 2 or more prior lines of therapy: the ROCKstar Study. Blood. 2021;138(22):2278-2289. doi:10.1182/blood.2021012021 3. Data on file 1. Kadmon Pharmaceuticals, LLC; 2021. 4. Lee SJ, Nguyen TD, Onstad L, et al. Success of immunosuppressive treatments in patients with chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2018;24(3):555-562. doi:10.1016/j.bbmt.2017.10.042 5. Data on file 2. Kadmon Pharmaceuticals, LLC; 2019. 6. Data on file 3. Kadmon Pharmaceuticals, LLC; 2021. 7. Data on file 4. Kadmon Pharmaceuticals, LLC; 2021. 8. Broadening the definition of tolerability in cancer clinical trials to better measure the patient experience. Friends of Cancer Research. Published October 24, 2018. Accessed April 19, 2022. https://www.focr.org/sites/default/files/Comparative Tolerability Whitepaper_FINAL.pdf